With silica stems and carbonate cones and corals all in a row.
______
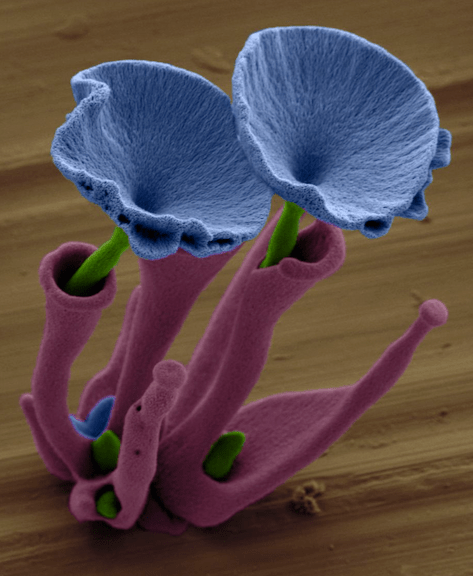
False-color electron micrograph* of a barium carbonate/silica ‘flower’ standing approximately 100 micrometers tall (courtesy of Wim Noorduin).
______
How do you sculpt a flower so small that even a honey-bee would never see it? There are ways to be sure, but they are either expensive or crude, and the complexity of a life-sized flower is still nearly impossible to mimic on a very small scale.
That may be about to change. In the cover article of tomorrow’s Science magazine, engineers in the laboratory of Joanna Aizenberg at Harvard University demonstrate that they can control the emergence of intricate structures standing less than one-tenth of a millimeter high.
The secret lies in the slow diffusion of carbon dioxide, or CO2, from the air into a water solution. The CO2 sets off a crystallization cascade of two simple materials: a metal carbonate, a similar material to the skeleton of marine corals; and, silicon dioxide, or silica, the primary component of sand and glass.†
The scientists have a number of tools with which to carve their micro-sculptures. “Since we have an understanding of the underlying mechanism in our system, we know how we can manipulate it to rationally get shapes that we want,” explains lead author and sculptor, Dr. Wim Noorduin. By modifying the temperature, the solution depth, the amount of CO2 they allow in, and the pH (a measure of acidity or basicity of the solution), they can precisely control the shapes that materialize—from stems, cones, and coral-shapes to helices and sprawling leaves.§
Remarkably, the scientists can even mold the structures as they grow by making simple changes to their mother solution. As Noorduin puts it, “You can really collaborate with the self-assembly process in time.”
They can alter the pH mid-growth to create helical stalks topped by blooms of corals and cones. Or, by controlling the influx of CO2, they can use the thickening and thinning of a structure’s walls to write messages in Morse code. They can make stems in vases topped by cones and coral blossoms, or corals with spiral arms that intertwine with their neighbors.
However, because an individual sculpture is so small, the exact chemistry surrounding it will be slightly different from its nearest neighbor, and this leaves each sculpture with a unique shape. “I think that is also why these structures look so organic,” says Noorduin. “It is so dependent on the local environment, and the local environment will be different everywhere.”
The scientists are now working on ways to control both this local environment and the types of materials that can be molded in this way. If they can, these structures may have applications beyond their artistic appeal, in fields from catalysis to microelectronics. “That would be of course nice,” muses Noorduin, “if you could get photovoltaic flowers.”
______
Electron micrograph of field of barium carbonate/silica stems, cones, and corals (courtesy of Wim Noorduin).
______
False-color electron micrograph of a strontium carbonate/silica ‘flower’ grown inside a barium carbonate and silica ‘vase’. The vase is grown first, the stem then nucleates and crystallizes from inside of it. A burst of CO2 causes the flower to ‘bloom’ (courtesy of Wim Noorduin).
______
Abraham Lincoln observes sculptures assembled on on the steps of the Lincoln Memorial–the Lincoln Memorial on the back of a penny, that is (courtesy of Wim Noorduin).
______
A field of barium carbonate/silica cones whose walls are thinned and thickened by alternating bursts of CO2 (courtesy of Wim Noorduin).
______
*These structures are so small that the only way to see them in detail is using an electron microscope.
†Their method works because as the metal carbonate crystallizes, the chemistry of the solution just around the edges of the crystal changes ever so slightly. This tiny change is just enough to set off the co-precipitation of the silica. Anywhere the silica grows, the metal carbonate crystal is stopped in its tracks. This means that the metal carbonate is in a race against time to grow away from the areas where the chemistry is right for silica crystallization.
§And what causes the different structure-types? At one pH (dubbed “the blossoming regime” by the authors), depending on how many neighbors it has, the sculpture will propagate in a straight line (creating stems), fan out from the original crystal (creating cones), or grow randomly in three dimensions (creating corals). At another (“the curling regime”), growing in on itself is the quickest way for a sculpture to escape the suffocating silica sheath. And so, it wraps around itself, around its nearest neighbor, or sticks close to the surface, creating single and double helices and flat planes.
More questions? Leave a comment!